Batteries are used to power toys, torches and other items that need energy to work.
Batteries store energy as chemical energy which is converted into electrical energy when the battery is part of a circuit.
When a battery is placed inside a circuit it pushes charge around the circuit. This pushing force is called voltage.
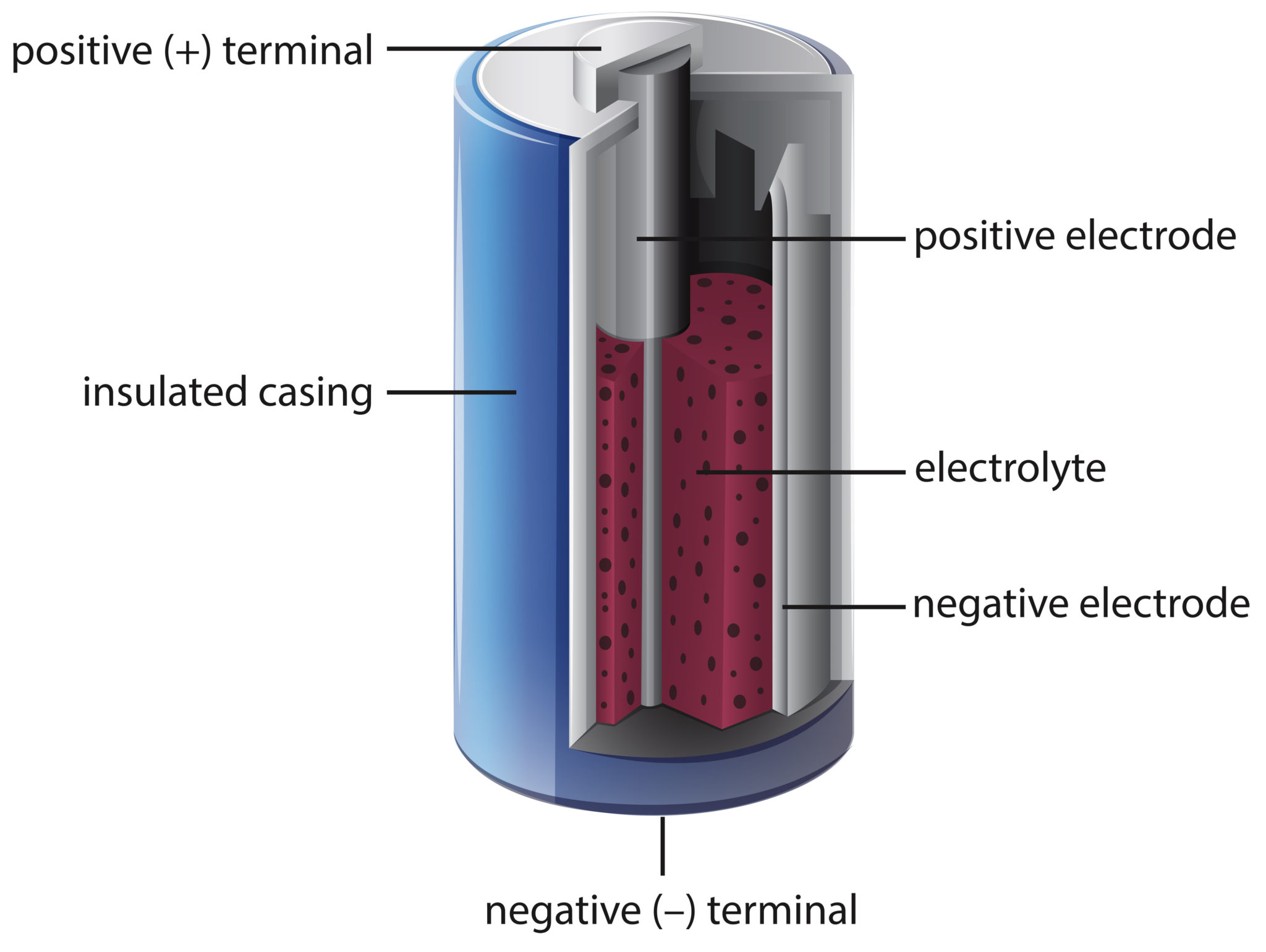
Batteries contain two different types of metal and an electrolyte.

Batteries contain three things:
- A positive electrode
- A negative electrode
- An electrolyte – a liquid or paste
Both electrodes have an end ( terminal ) that extends out of the battery.
Make a lemon battery
You can make a very simple battery with a lemon, copper wire or coin and a zinc ( galvanised ) nail. The lemon juice, copper and zinc react with each other creating electricity!

Electrons build up on the zinc nail and leave the copper coin or wire. The electrons want to move to the copper coin but can’t travel through the electrolyte. When a wire is connected between the nail and coin electrons flow down the wire between the nail and copper coin. If an LED is placed in the circuit it will light up as current passes through ( current is electrons flowing through the wire ).

What’s in a dry battery?
Real batteries aren’t made with lemons but have a metal cap which acts as the positive electrode attached to a carbon rod and a negative electrode made from zinc. The electrolyte is mixed with manganese dioxide making it solid. This type of battery is called a dry battery or cell.
A battery that is only used once is a primary battery, this is the type of battery is commonly used in remote controls and other small items. Batteries that can be recharged are secondary batteries ( examples of these are mobile phones and car batteries ).
Remember
Electric current is the flow of electrons around a circuit. If the circuit breaks the current cannot flow.
More circuit investigation ideas
Create a circuit with graphite!
Make a potato battery!
Find out what happens when you break a circuit by adding a switch.

The post How do batteries work? appeared first on Science Experiments for Kids.



